Shrinking the carbon footprint of chemical manufacturing with lasers, solar radiation – University of Illinois Urbana-Champaign
Innovative Solar-Powered Chemical Reaction Advances Sustainable Manufacturing
Introduction
Researchers at the University of Illinois Urbana-Champaign have developed a novel method to harness solar energy for driving olefin epoxidation, a critical chemical reaction widely used in manufacturing industries such as textiles, plastics, chemicals, and pharmaceuticals. This breakthrough aligns with several Sustainable Development Goals (SDGs), particularly SDG 7 (Affordable and Clean Energy), SDG 9 (Industry, Innovation, and Infrastructure), and SDG 13 (Climate Action), by reducing energy consumption, eliminating harmful byproducts, and minimizing carbon emissions.
Background: Challenges in Olefin Epoxidation
- Olefin epoxidation produces epoxide chemicals essential for multiple industries.
- Current industrial processes rely on harsh peroxides that are difficult to dispose of safely and generate carbon dioxide emissions.
- Using water as an oxidant is environmentally preferable but requires high temperatures to break strong H–O–H bonds, leading to high energy use and increased CO2 emissions.
A greener alternative is necessary to significantly reduce the chemical manufacturing industry’s carbon footprint, supporting SDG 12 (Responsible Consumption and Production).
Research Innovation: Plasmonic Chemistry Using Solar Energy
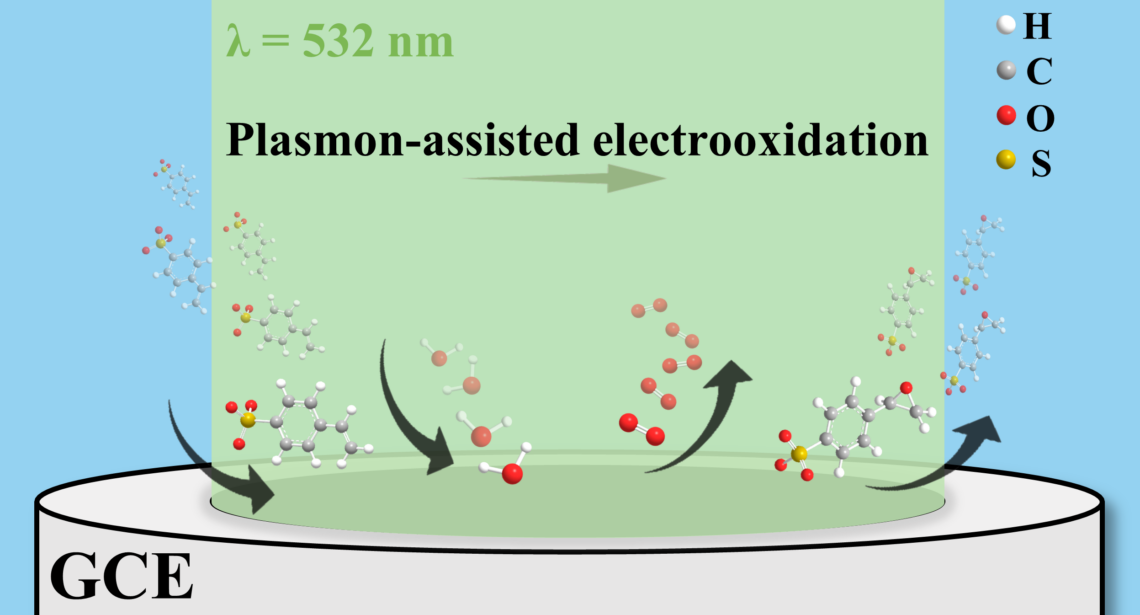
Professor Prashant Jain’s research group specializes in plasmonic chemistry, a process that uses solar energy to enhance chemical reactions. Their recent study, published in the Journal of the American Chemical Society, demonstrates the application of this technique to epoxidation reactions, potentially revolutionizing chemical manufacturing and electrochemistry.
Key Features of the New Method
- Use of light-absorbing “antenna” catalysts composed of gold nanoparticles and manganese oxide nanowire electrodes.
- Combination of electrical energy and visible-light photons to break water’s H–O–H bonds at ambient temperature.
- Elimination of the need for high-temperature heating, reducing energy consumption and carbon emissions.
Mechanism of Action
Visible light photons from laboratory lasers are absorbed by the nanoparticles, generating strong electric fields and energetic charge carriers. These weaken the O–H bonds in water and the double bonds in styrene, enabling oxygen atoms to be extracted from water and incorporated into epoxide molecules through a light-catalyzed reaction.
Implications for Sustainable Development
- SDG 7 (Affordable and Clean Energy): Utilizes solar energy to drive chemical reactions, reducing reliance on fossil fuels.
- SDG 9 (Industry, Innovation, and Infrastructure): Introduces innovative catalytic technology that can transform industrial chemical processes.
- SDG 12 (Responsible Consumption and Production): Minimizes hazardous waste by replacing harsh peroxides with water as an oxidant.
- SDG 13 (Climate Action): Lowers carbon emissions associated with chemical manufacturing.
Challenges and Future Directions
While the laboratory-scale demonstration is promising, scaling this technology for industrial application presents challenges:
- Replacing laboratory lasers with scalable, energy-efficient light sources.
- Enhancing control over light-driven reactions to prevent overoxidation.
- Engineering large-scale, light-accessible electrolyzer systems to replicate lab-scale efficiency.
Funding and Collaborations
This research was supported by the National Science Foundation, the São Paulo Research Foundation, and the U.S. Department of Energy. Collaborators include Susana Inés Córdoba de Torresi from the Universidade de São Paulo and George Schatz from Northwestern University.
Contact Information and Access to Publication
- Contact: Professor Prashant Jain
- Phone: 217-333-3417
- Email: jain@illinois.edu
- Research Paper: Plasmon-assisted electrochemical epoxidation using water as an oxidant
1. Sustainable Development Goals (SDGs) Addressed or Connected
- SDG 7: Affordable and Clean Energy
- The article discusses using solar energy and visible light photons to power chemical reactions, promoting renewable energy use.
- SDG 9: Industry, Innovation and Infrastructure
- The research advances industrial chemical manufacturing by introducing greener, energy-efficient processes.
- SDG 12: Responsible Consumption and Production
- The new method reduces harsh oxidizing byproducts and carbon emissions, promoting sustainable industrial processes.
- SDG 13: Climate Action
- Minimizing carbon emissions in chemical manufacturing contributes to climate change mitigation.
2. Specific Targets Under Those SDGs Identified
- SDG 7: Affordable and Clean Energy
- Target 7.2: Increase substantially the share of renewable energy in the global energy mix.
- SDG 9: Industry, Innovation and Infrastructure
- Target 9.4: Upgrade infrastructure and retrofit industries to make them sustainable, with increased resource-use efficiency and greater adoption of clean and environmentally sound technologies.
- SDG 12: Responsible Consumption and Production
- Target 12.4: Achieve the environmentally sound management of chemicals and all wastes throughout their life cycle.
- Target 12.5: Substantially reduce waste generation through prevention, reduction, recycling and reuse.
- SDG 13: Climate Action
- Target 13.2: Integrate climate change measures into national policies, strategies and planning.
3. Indicators Mentioned or Implied to Measure Progress
- Indicator for SDG 7.2:
- Proportion of renewable energy in total final energy consumption — implied by the use of solar energy and visible light photons to power chemical reactions.
- Indicator for SDG 9.4:
- CO2 emission per unit of value added — implied by the reduction of carbon emissions in chemical manufacturing processes.
- Adoption rate of clean and environmentally sound technologies in industry — implied by the introduction of plasmonic chemistry and light-driven electrochemical processes.
- Indicators for SDG 12.4 and 12.5:
- Amount of hazardous waste generated and managed safely — implied by elimination of harsh oxidizing byproducts and safer oxidants.
- Waste generation per unit of production — implied by reduction of harmful chemical waste.
- Indicator for SDG 13.2:
- Number of policies integrating climate change measures — implied by research contributing to climate action through cleaner industrial processes.
4. Table of SDGs, Targets, and Indicators
| SDGs | Targets | Indicators |
|---|---|---|
| SDG 7: Affordable and Clean Energy | 7.2: Increase substantially the share of renewable energy in the global energy mix. | Proportion of renewable energy in total final energy consumption (implied by solar energy use). |
| SDG 9: Industry, Innovation and Infrastructure | 9.4: Upgrade infrastructure and retrofit industries to make them sustainable, with increased resource-use efficiency and greater adoption of clean technologies. | CO2 emission per unit of value added; Adoption rate of clean and environmentally sound technologies (implied by plasmonic chemistry application). |
| SDG 12: Responsible Consumption and Production | 12.4: Achieve environmentally sound management of chemicals and wastes. 12.5: Substantially reduce waste generation. |
Amount of hazardous waste generated and managed safely; Waste generation per unit of production (implied by elimination of harsh oxidizing byproducts). |
| SDG 13: Climate Action | 13.2: Integrate climate change measures into national policies, strategies and planning. | Number of policies integrating climate change measures (implied by research supporting cleaner industrial processes). |
Source: news.illinois.edu
What is Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0